| GEOLOGICAL SYSTEM
Mechanisms of Generation, Migration Pathways, and Models of Accumulation and Loss of Natural Hydrogen.
Serpentinization
Water infiltrates ultramafic rocks through fractures, reacting with present minerals and releasing hydrogen. This process involves the oxidation of iron and the reduction of water.
Settings: Ultramafic Rocks, Oceanic Ridges, Ophiolites.
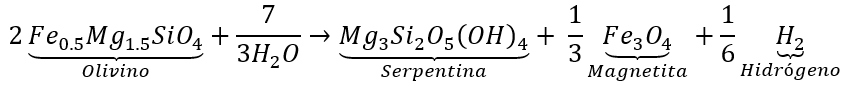 Specific reaction of hydrogen generation by serpentinization.
Specific reaction of hydrogen generation by serpentinization. Source: (Marcaillou et al., 2011).
Water Radiolysis
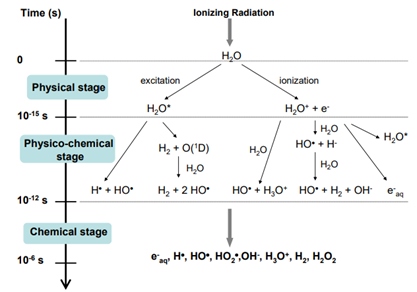 Main reactions in the three stages of water radiolysis.
Main reactions in the three stages of water radiolysis. Source: Le Caër, 2011.
The radioactive decay of elements such as U, Th, and K in rocks emits ionizing radiation that dissociates water molecules, generating free radicals. Their recombination produces molecular hydrogen.
Settings: Type A Granites, Cratons, Precambrian Shields, Rocks with high radioactivity.
Mechanoradicals
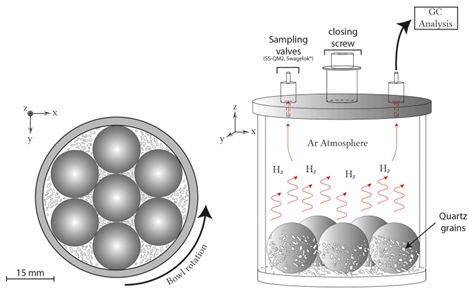 Grinding scheme used to study hydrogen generation during quartz crushing.
Grinding scheme used to study hydrogen generation during quartz crushing. Source: Lefeuvre et al., 2025.
During tectonic faulting, mineral fractures generate fresh surfaces with highly reactive free radicals. These radicals interact with water molecules (H₂O), promoting redox reactions that release molecular hydrogen.
Settings: Tectonically active zones.
Magmatic Degassing
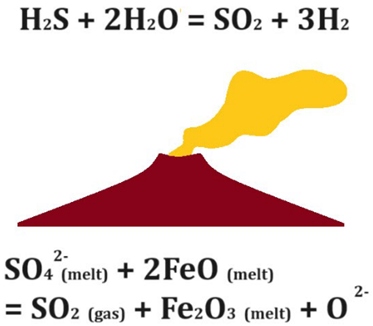 Hydrogen generation through magmatic degassing.
Hydrogen generation through magmatic degassing. Source: (Feodoridi & Feodoridi, 2024).
In volcanic zones, in addition to water vapor and CO₂, hydrogen is also released. This gas may come directly from magma or form when compounds like hydrogen sulfide (H₂S) react with water or minerals at high temperatures, especially in hydrothermal systems.
Settings: Fumaroles, Volcanic Zones.
Oxidation of Fe²⁺ in Silicate Minerals
Fe²⁺ in biotite or hornblende is oxidized during hydrothermal processes, releasing electrons that reduce protons in water, generating molecular hydrogen and secondary minerals such as hematite or magnetite.
Settings: Altered granites, Hydrothermal zones.
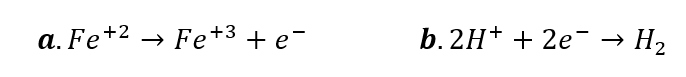 (a) Ferrous iron (Fe²⁺) oxidizes to ferric (Fe³⁺), releasing electrons. (b) Electrons reduce protons (H⁺) to form molecular hydrogen.
(a) Ferrous iron (Fe²⁺) oxidizes to ferric (Fe³⁺), releasing electrons. (b) Electrons reduce protons (H⁺) to form molecular hydrogen. Source: (Murray et al., 2020).
Martitization
Magnetite (Fe₃O₄) transforms into hematite (Fe₂O₃) under oxidizing conditions in the presence of water. Fe²⁺ in magnetite oxidizes to Fe³⁺, which incorporates into hematite, releasing electrons that reduce water protons to generate hydrogen (Geymond et al., 2022).
Settings: Banded Iron Formations (BIF), Cumulate Gabbros.
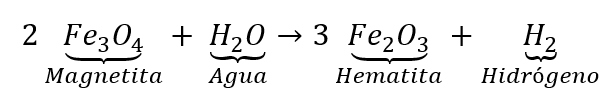 Reaction for hydrogen generation via martitization.
Reaction for hydrogen generation via martitization. Source: (Feodoridi & Feodoridi, 2024).
Organic Matter Pyrolysis
 Hydrocarbon model with hydrogen addition.
Hydrocarbon model with hydrogen addition. Source: (Hanson & Hanson, 2024).
A thermochemical process where kerogen-rich organic matter decomposes under high temperature (>300 °C) and pressure. At temperatures of 300–600 °C or more, molecular hydrogen and carbon (graphite) are formed (CH₄ = C + 2H₂).
Settings: Deep sedimentary basins with high thermal maturity, Magmatic-influenced areas.
Loss Mechanisms
Hydrogen can migrate through geological structures such as faults and fractures, and also diffuse slowly through rock matrices. This phenomenon may result in surface features like "fairy circles.".
Near-surface subsurface microorganisms can consume hydrogen as an energy source, producing gases like methane (CH₄) as byproducts.
In deeper zones, hydrogen reacts with minerals and other gases, forming H₂O, CH₄, and various mineral compounds via abiotic reactions.
Conceptual models of the hydrogen system
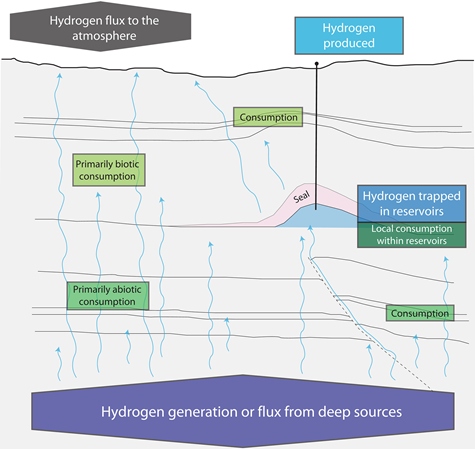 Conceptual model of geological hydrogen resources.
Conceptual model of geological hydrogen resources. Source: (Ellis & Gelman, 2024).
Hydrogen is generated at depth, migrates, is consumed (biotically or abiotically), accumulates in geological traps, or escapes to the atmosphere.
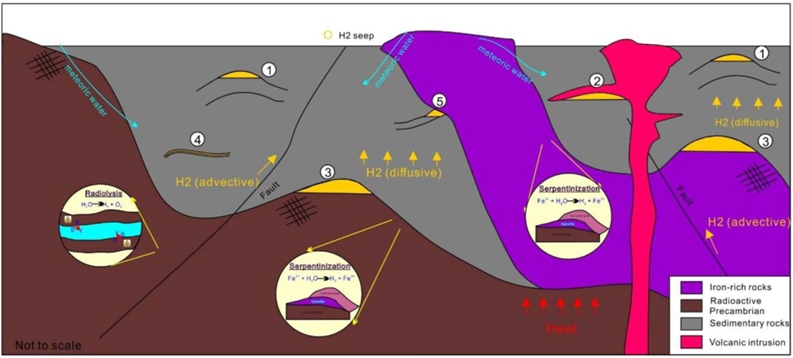 Schematic diagram showing a shallow hydrogen system model with potential trap types: structural trap, non-sedimentary seal structural trap, non-sedimentary reservoir trap, adsorbed hydrogen, and stratigraphic trap. Two iron-rich rock types are included: Precambrian craton and ophiolite belts, which act as the main natural hydrogen source rocks. Heat sources (e.g., magmatism) will enhance hydrogen generation.
Schematic diagram showing a shallow hydrogen system model with potential trap types: structural trap, non-sedimentary seal structural trap, non-sedimentary reservoir trap, adsorbed hydrogen, and stratigraphic trap. Two iron-rich rock types are included: Precambrian craton and ophiolite belts, which act as the main natural hydrogen source rocks. Heat sources (e.g., magmatism) will enhance hydrogen generation. Source: (Patiño et al., 2024).
A shallow system where iron-rich rocks (ophiolites or Precambrian) generate hydrogen via serpentinization and radiolysis at < 100 °C. Hydrogen migrates by diffusion or through fractures and accumulates in structural, stratigraphic, or adsorbed clay traps. Groundwater and magmatic heat can enhance the system.
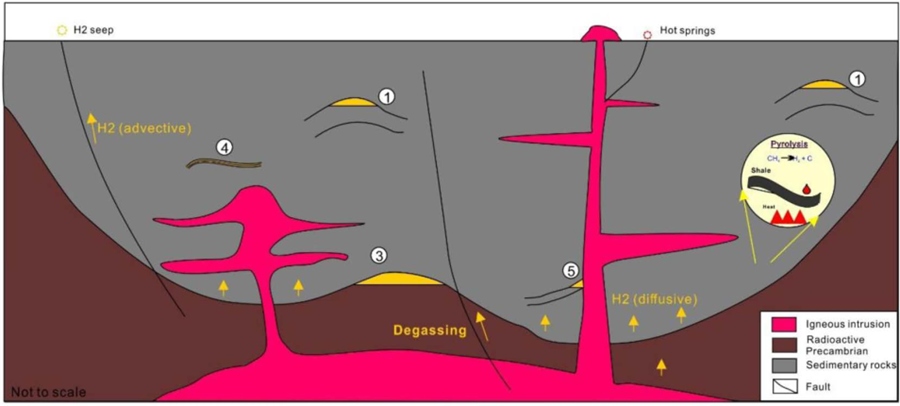 Schematic diagram showing a model of a deep hydrogen system with possible trap types. The high temperature caused by deep burial and/or thermal events make pyrolysis a possible hydrogen generation mechanism.
Schematic diagram showing a model of a deep hydrogen system with possible trap types. The high temperature caused by deep burial and/or thermal events make pyrolysis a possible hydrogen generation mechanism. Source: (Zhao et al., 2023).
Hydrogen is generated through organic matter pyrolysis (>300 °C) and mantle degassing, typically in rift basins with high thermal gradients and deep faults acting as conduits. Gas accumulates in traps under favorable geological conditions.
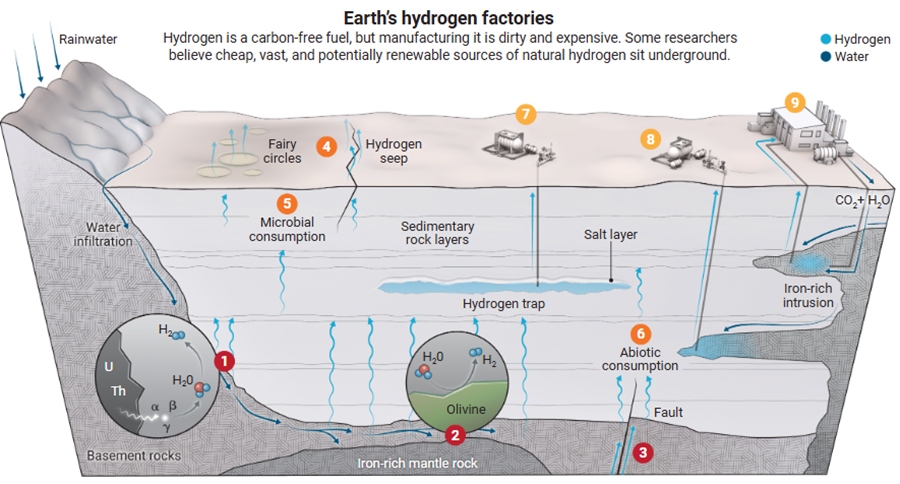 Conceptual model of hydrogen generation, loss, and extraction in the subsurface.
Conceptual model of hydrogen generation, loss, and extraction in the subsurface. Source: (Hand, 2023).
Geological hydrogen is generated via water radiolysis, serpentinization, or deep mantle flows. Losses occur through leakage, microbial consumption, or rock interactions. Extraction is possible via drilling traps, accessing iron-rich rocks, or stimulating production (e.g., water/CO₂ injection for co-capture).
Comparison Between H₂ and HC Systems
Although natural hydrogen accumulation mechanisms are not yet fully understood, evidence from Bourakebougou suggests the presence of generation, migration, and trapping systems similar to petroleum systems, though with fundamental differences.
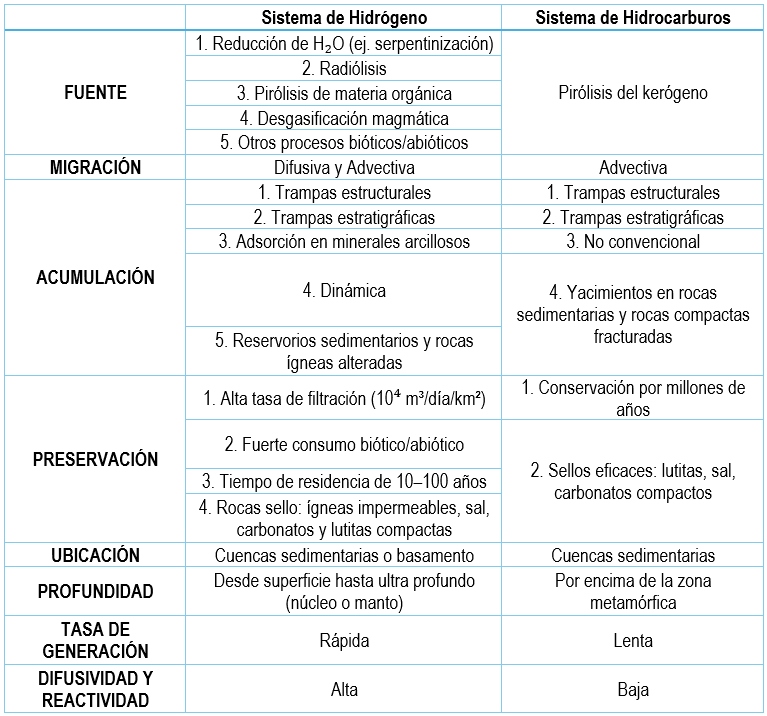 Comparison between the Hydrogen System and the Hydrocarbon System.
Comparison between the Hydrogen System and the Hydrocarbon System. Modified from: (Zhang & Li, 2024, Zhao et al., 2023).
Bibliographic References
- Ellis, G. S. & Gelman, S. E. (2024). Model predictions of global geologic hydrogen resources. Sci. Adv. 10 , eado0955 - Check this reference here
- Feodoridi, K., & Feodoridi, G. (2024). Geological hydrogen. Different sources and mechanisms for the production of natural hydrogen. Preprint, 1–25. - Check this reference here
- Geymond, U., Ramanaidou, E., Lévy, D., Ouaya, A., & Moretti, I. (2022). Can Weathering of Banded Iron Formations Generate Natural Hydrogen? Evidence from Australia, Brazil and South Africa. Minerals, 12(2), 163. - Check this reference here
- Hand, E. (2023). Hidden Hydrogen: Does Earth hold vast stores of a renewable, carbon-free fuel? Science, 379(6633), 630–636. - Check this reference here
- Hanson, J., & Hanson, H. (2024). Hydrogen’s organic genesis. Unconventional Resources, 4, 100057. - Check this reference here
- Le Caër, S. (2011). Water Radiolysis: Influence of Oxide Surfaces on H2 Production under Ionizing Radiation. Water, 3(1), 235-253. - Check this reference here
- Lefeuvre, N., Truche, L., Donzé, F.-V., Vandenborre, J., Gaucher, E. C., & Magnin, V. (2025). The contribution of mechanoradical reactions to crustal hydrogen generation. Earth and Planetary Science Letters, 660, 119363. - Check this reference here
- Marcaillou, C., Muñoz, M., Vidal, O., Parra, T., & Harfouche, M. (2011). Mineralogical evidence for H2 degassing during serpentinization at 300°C/300bar. Earth and Planetary Science Letters, 303(3–4), 281–290. - Check this reference here
- Zhang, M, & Li Y. (2024). The role of geophysics in geologic hydrogen resources. Journal of Geophysics and Engineering (2024) 21, 1242–1253. - Check this reference here
- Zhao, H., Jones, E. A., Singh, R. S., Ismail, H. H. B., & Tan, S. W. (2023). The hydrogen system in the subsurface: Implications for natural hydrogen exploration. Paper presented at the ADIPEC, Abu Dhabi, UAE. Society of Petroleum Engineers. SPE-216710-MS. - Check this reference here